
Covalent Bonds Biology for Majors I
Dichlorine monoxide (OCl2) is an inorganic compound that is brown-yellow gas at room temperature. It has a molecular weight of 86.9054 g/mol. It has a melting point of -120.6℃ and a boiling point of 2.0℃. Belonging to the chlorine oxide family, OCl2 is soluble in both water and inorganic solvents.
Ppt Polar Bonds And Molecules Powerpoint Presentation, Free Download FD6
Question: and VSEPR structure for OCl2 A. Give the electron geometry, (Select] B. Give the bond angle. (Select) C. Give the molecular geometry (Select) D. Does it have polar covalent bonds? (Select) E Is the molecule polar or nonpolar? [Select) F. List all the intermolecular forces present. (Select) G. Give the strongest intermolecular force.

Is OCl2 Polar or Nonpolar (Dichlorine Monoxide) YouTube
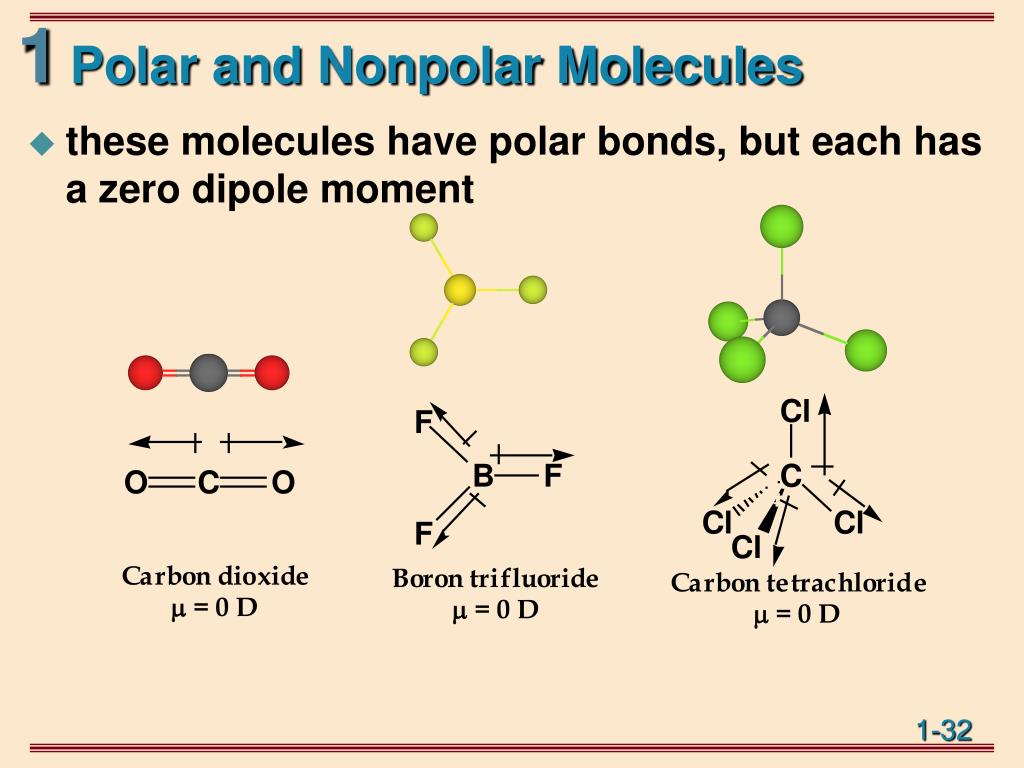
Each C-O bond in CO 2 is polar, yet experiments show that the CO 2 molecule has no dipole moment. Because the two C-O bond dipoles in CO 2 are equal in magnitude and oriented at 180° to each other, they cancel. As a result, the CO 2 molecule has no net dipole moment even though it has a substantial separation of charge.
How To Know If A Molecule Is Polar Or Nonpolar Khan Academy
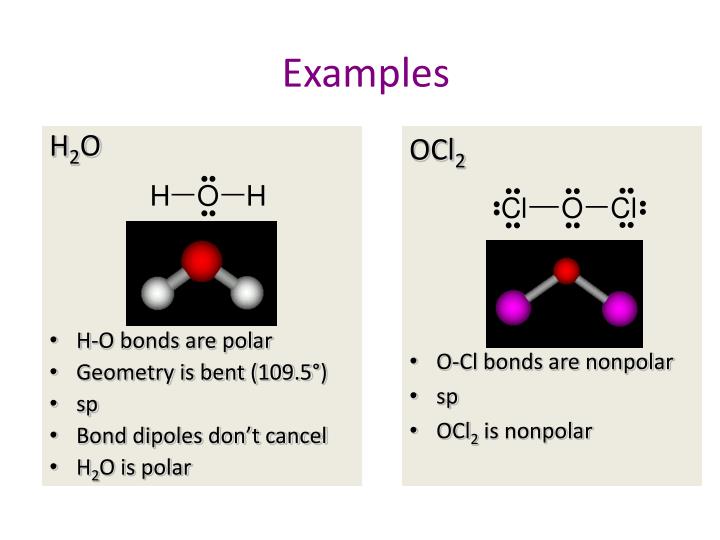
Is the molecule O C l 2 polar or nonpolar? Explain. Dichlorine Monoxide: Dichlorine monoxide, also called chlorine monoxide, is a brownish-yellow color gas. It is soluble in water and in.

Is OCl2 Polar or Nonpolar? Techiescientist
2.45K subscribers 1.7K views 1 year ago Polarity of Molecules OCl2 is a chemical formula for Oxygen Chloride. To find out whether the molecule is polar or nonpolar, we will first look at the.

How Do You Teach Polar Vs. Nonpolar Molecules? — CoScine Creative
This article explain drawing Cl 2 O Lewis structure, shape, formal charge, resonance in Cl 2 O. As next part, the bond angle, octet rule, Cl 2 O Lewis structure lone pairs of Valence electron, covalent nature, hybridization, solubility, and Cl 2 O polarity also being elaborated.. Dichlorine monoxide is the chemical name for Cl 2 O. Oxygen atom has got two chlorine atoms around it in Cl 2 O.

PPT Lewis Structures PowerPoint Presentation ID5585056
Dichlorine monoxide is an inorganic compound with the molecular formula Cl 2 O. It was first synthesised in 1834 by Antoine Jérôme Balard, [2] who along with Gay-Lussac also determined its composition.
PPT Lewis Structures PowerPoint Presentation ID5585056
Is OCl2 polar or nonpolar? Question = Is OCl2 polar or nonpolar? Answer = OCl2 is Polar What is polar and non-polar? Polar "In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment.
[Solved] image attached 1. Complete the table below. Indicate whether
By Triyasha Mondal Ocl2 lewis structure shows the bonding structure of involving atoms in the molecule. The article will discuss about it briefly. Outer shell electrons of the involved atoms are shown in the ocl2 lewis structure. These electrons effect the properties of the molecule.
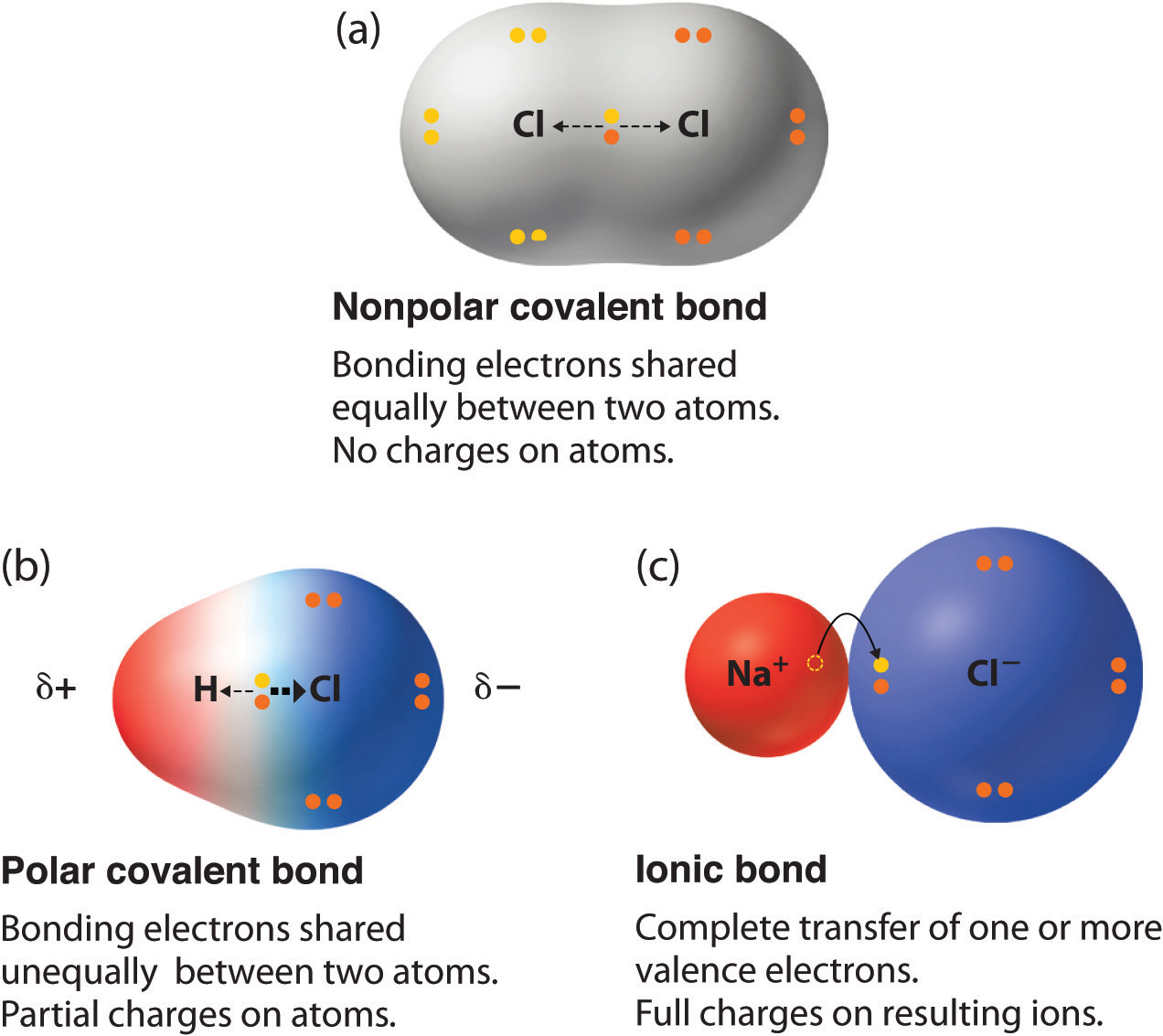
Polar vs Nonpolar bonds What is the Main Difference? PSIBERG
Explain how a molecule that contains polar bonds can be nonpolar. Answer. As long as the polar bonds are compensated (for example. two identical atoms are found directly across the central atom from one another), the molecule can be nonpolar. PROBLEM \(\PageIndex{2}\)

Polar vs. Nonpolar Bonds — Overview & Examples Expii Ionic Bonding
OCL2, or chlorine (I) oxide, is a chemical compound composed of 1 chlorine atom and one oxygen atom. Its chemical component is Cl2O, a yellowish-inexperienced gas at room temperature. The molecule has a dishonest shape, resulting in its polar nature.

Lewis Structure For Ocl2
(And Why?) May 25, 2023 by Jay Rana OCl2 is a POLAR molecule. But why? And how can you say that OCl2 is a polar molecule? Want to know the reason? Let's dive into it! OCl2 is a POLAR molecule because it has a lone pair of electrons on the Oxygen atom (O) which causes the entire molecule to bend.
Polar and Nonpolar Covalent Bonds Characteristics & Differences
Oxygen dichloride has the chemical formula OCl 2 with a molar mass of 86.9054 g/mol. It appears as brownish-yellow gas. It is soluble in water. In this article, we will discuss OCl 2 lewis structure, molecular geometry, bond angle, polar or nonpolar, its hybridization, etc. Oxygen dichloride is a member of the chlorine oxide family of compounds.
Cómo saber si una molécula es polar o no polar La Ciencia y la
Science Chemistry Chemistry questions and answers Determine whether the following are polar or non-polar: 1. CF3Cl : polar 2. C2H2 : non-polar 3. SF2Cl2 : polar 4. H2O2 : non-polar 5. OCl2 : polar 6. PCl4 : non-polar 7. PO43− : non-polar This problem has been solved!

Is SF4 polar or nonpolar [1 Best explanation] Science Education and
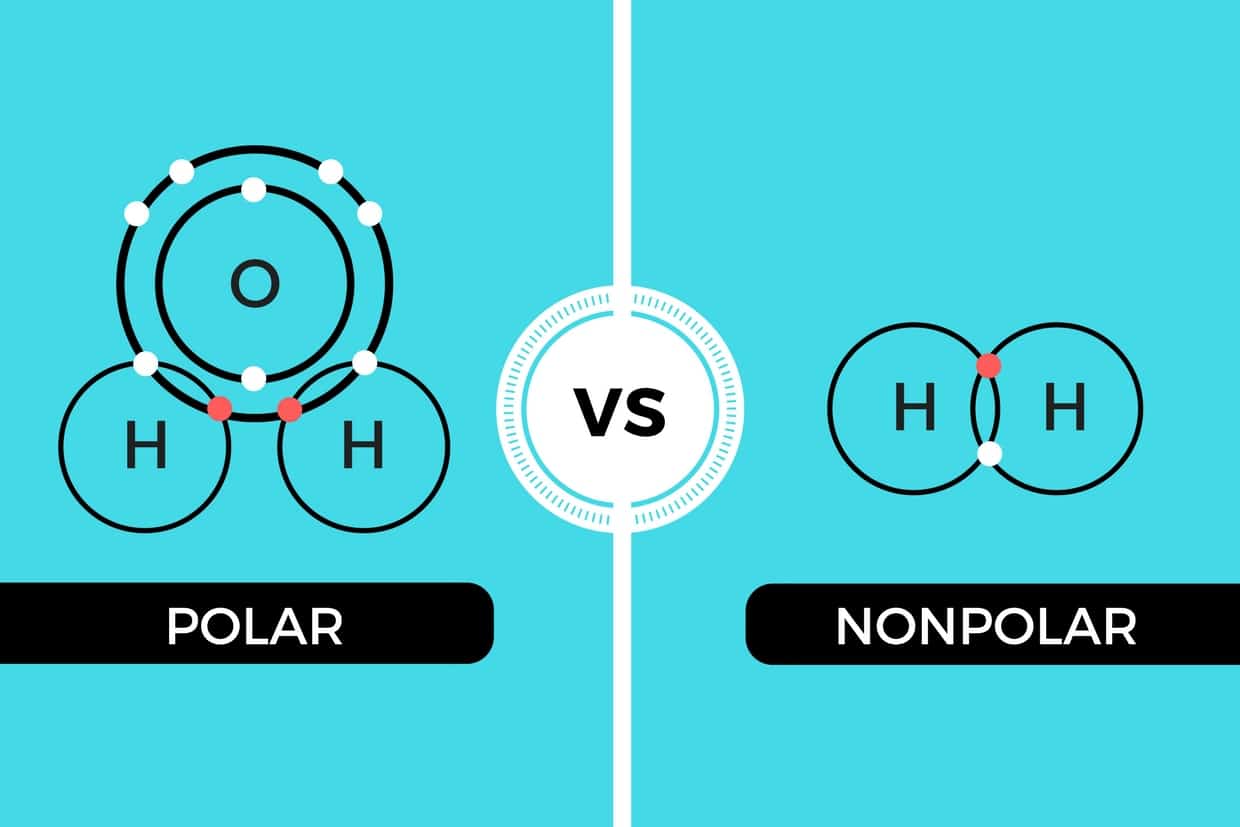
The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.
Ms J's Chemistry Class Polar vs NonPolar Covalent Bonds
Expert Solution Trending now This is a popular solution! Step by step Solved in 2 steps with 1 images SEE SOLUTION Check out a sample Q&A here Knowledge Booster Learn more about Theories of Bonding Need a deep-dive on the concept behind this application? Look no further.